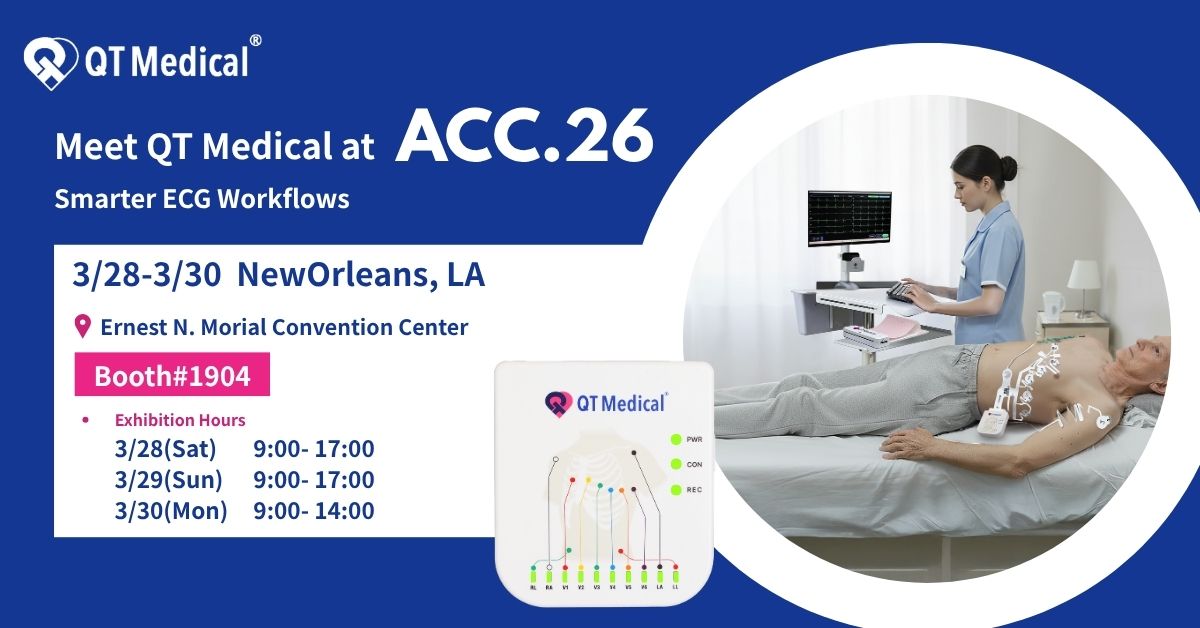
News
Total
26
-
-
Los Angeles, CA, December 22, 2025Happy Holidays from QT Medical.As the year wraps up, we’re grateful for the conversations, shared challenges, and moments of progress we experienced together.
-
Los Angeles, CA –, August 29, 2025QT Medical, a pioneer in hospital-grade, portable 12-lead electrocardiogram (ECG) solutions, is honored to be named a finalist in the Best Innovation in Medical Technology – Device category at the 2025 Octane OC High Tech Awards. H
-
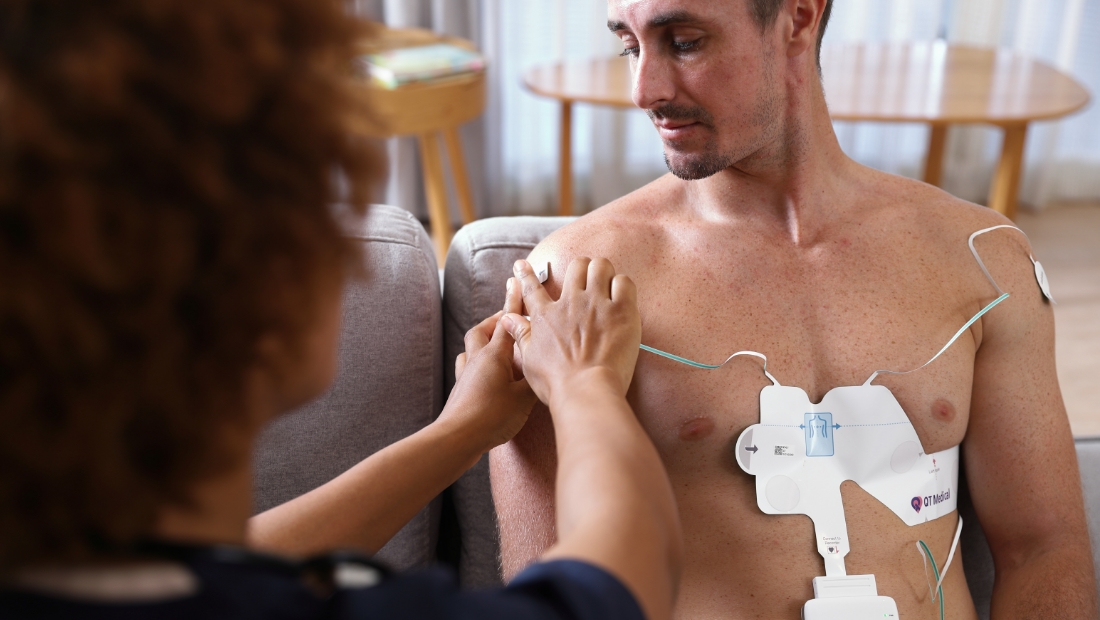
Dallas, TX –, August 28, 2025QT Medical, Inc. has focused on making hospital-grade PCA 500 12-lead ECG System more accessible outside traditional clinical settings. The platform is a compact, wireless, FDA-cleared system suitable for home care, telehealth, clinical trials, rural health, and conventional hospitals.
-
location test3, March 18, 2025As a pioneer in ECG solutions, QT Medical offers its flagship product, PCA 500, a compact, digital, and wireless 12-lead ECG system designed for both professional and personal use.
-
LOS ANGELES, CA, UNITED STATES, March 11, 2025QT Medical, a leader in innovative ECG solutions, proudly announces that its flagship product, PCA 500, has received Health Canada registration, marking a significant milestone in the company’s global expansion.